Post
Steady State
8 December 2012
Yesterday I wrote about radioactive decay and how we can use the half-life property of radioactive isotopes to determine the age of things. Today we’ll see how this applies to a bit of astrophysics.
Because radioactive isotopes decay, the amount of that isotope will decrease over time unless it is replenished by some type of mechanism. For carbon-14 that mechanism is cosmic rays striking the upper atmosphere. When there is a replenishing mechanism then the amount of isotope will reach a stable level where the rate at which new isotopes are created are balanced by the rate at which the isotopes decay. Given the half-life of an isotope we can calculate the rate at which the isotope is decaying, and therefore we can determine the rate at which new isotopes are being created.
 Max-Planck-Instituts für extraterrestrische Physik
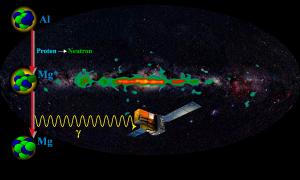
Max-Planck-Instituts für extraterrestrische PhysikThere are lots of radioactive isotopes out in the galaxy, but one of these isotopes, aluminum-26 tells us something interesting about supernovae. When aluminum-26 decays into magnesium it gives off light (gamma rays) at a particular wavelength. By measuring the brightness of light we can determine the amount of aluminum-26 in our galaxy. What we find is that there is much more aluminum-26 in our galaxy than we would expect, which means something must be replenishing it. This is interesting because the primary source of aluminum-26 is supernovae. When a supernova explodes it gives of lots of radioactive isotopes, and aluminum-26 is one of them.
Since we know how much aluminum-26 is in our galaxy, we know the rate at which it is being replenished by supernovae. So we can determine the rate at which supernovae occur in our galaxy. What we find is that we typically have a supernova about once every 50 years. This rate is consistent with the rate we see in similar galaxies.