Post
A Muse of Fire
26 March 2013
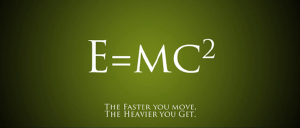 via Reddit
via RedditIn the mid-1800s, astronomy faced a serious problem. No one knew how the stars shone. Now a little mystery never hurt anyone, but in this case it was deeply perplexing. By this time we had a solid understanding of energy, and that energy is conserved. That is, energy must come from somewhere, and it must go to somewhere. We also had a general understanding of thermodynamics, specifically that you could squeeze something to heat it up. This gave us a mechanism which could cause the stars to shine, known as the Kelvin-Helmholtz mechanism.
The basic idea of this mechanism is that for a large body like a planet or star, gravity tries to compress the star or planet smaller and smaller. This compression heats the core of the body, which is radiated as light. In this way a star could be heated by its own weight. This is a very real effect for gas planets and brown dwarfs. Jupiter, for example, radiates more heat than it receives from the Sun because of this mechanism.
But for a star such as the Sun there is a problem. Gravity can only squeeze a body so far, so there’s a limit to how much heat can be generated by the Kelvin-Helmholtz mechanism. And by conservation of energy, once the Sun has radiated all its heat energy as light, it is done shining. It’s fairly easy to estimate the rate at which the Sun loses energy given its brightness and size. Its also fairly straightforward to calculate just how much energy the Sun could gain by gravitational compression. If you apply conservation of energy, then you can determine how long the sun could shine before it runs out of energy, and you get a clear answer: about twenty million years.
That’s quite a long time, but it disagreed horribly with geology, where fossil evidence demonstrated that life existed on Earth for several hundred million years, likely much longer. How is that possible if the Sun could only be tens of millions of years old? Kelvin actually argued that the geologists must be wrong, and the Earth was only millions of years old. But by the early 1900s, we understood that life on Earth was around not only millions, but billions of years. So the Sun must have shone for billions of years, but astronomers and physicists had no explanation for how that was possible. Gravitational compression couldn’t provide enough energy, nor could chemical reactions, and what else was there?
Then in 1905 Albert Einstein published his paper on special relativity. Usually when people talk about relativity they mention that time slows down at high speeds, or as the figure above claims the faster you move the more mass you have (which isn’t technically true, but I’ll explain that in a couple days). But central to all this wibbly-wobbly timey-wimey stuff is that energy and mass are two sides of the same coin. They are connected. Not only that, mass can become energy and energy can become mass. This connection is summarized in equation above. In the equation E stands for energy, m for mass, and c for the speed of light. What the equation says is that given a certain amount of mass, it is possible in principle to convert it into a certain amount of energy. Just how much energy you can get is surprisingly huge. The speed of light is about 300 million meters per second, and it is squared in the equation. To give you an idea of just how big this is, if we could convert one paper clip entirely to energy, it would produce enough electrical energy to power the entire world for about 800 billion years.
Einstein gave us the key to understanding the stars. Somehow the Sun was converting a bit of its mass into energy, and with it’s great mass the Sun could shine for billions upon billions of years. But while special relativity showed it was possible, it gave no clue as to how it actually occurred. There were, however, tantalizing clues. By the late 1800s Marie Curie and others had begun to study radioactive decay. By the early 1900s it was demonstrated that radioactive elements could transmute into other elements, which gave us another key to the puzzle. For example, through radioactive decay, a thorium atom can break apart into a radium atom and a helium atom (also known as an alpha particle). The mass of the radium and helium atoms is less than the mass of the original thorium, and the “missing mass” is converted to energy, as per Einstein’s equation above.
But this fission process only works for heavy elements. The sun and stars are mostly made of the light elements hydrogen and helium, and those can’t be split into lighter elements. But perhaps the tremendous heat and pressure of a sun’s core could fuse lighter elements into heavier ones. In 1939, Hans Bethe demonstrated how four hydrogen atoms could be fused into a single helium atom. A helium atom has less mass than four hydrogen, so the result was helium plus energy. We then learned how helium could become carbon, nitrogen and oxygen, and on to heavier and heavier elements.
This gave us far more answers than we expected. It not only gave us fusion as the source of a star’s power, but explained from whence the diversity of elements came. Hydrogen and helium fusing into heavier elements. Exploding stars scattering those elements across the cosmos. New stars forming, with planets, one of which is home to us. From a single, simple equation we came to know that we are the dust of stars.
Of course this equation is also a stark reminder that knowledge can be double-edged sword. In understanding the source of the Sun’s power, we longed to wield that power ourselves. By the mid-twentieth century we succeeded. We can now drop the heart of a star over the cities of our enemies, or power the probes that travel to the furthest reaches of our solar system and beyond.
Which muse we follow is up to us.
Coming tomorrow: How Newton united physics and astronomy, and brought us face to face with one of the most mysterious and terrifying objects in the universe. All in part two of this six part series.