Post
Back of the Envelope
22 February 2012
Astrophysics (like any science) is about getting the details right. As I’ve mentioned earlier, the difference between two theoretical models can be very small (look at the precession of Mercury as a case in point). But there are times when you just do a rough calculation to see if you are on the right track.
 Brian Koberlein
Brian KoberleinSuppose you wanted to determine the pressure and temperature inside the Sun. We could start with a very simple model: assume the Sun is an ideal gas, sitting there quietly under its own weight (known as hydrostatic equilibrium). You would think this is a horrible approximation. The Sun is nothing like an ideal gas, right? It certainly isn’t static. It has flares and convection and is fusing elements in its core. Still, let’s see what our simple model predicts.
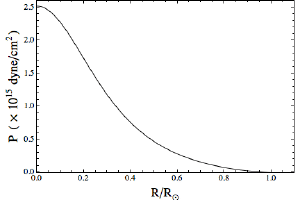
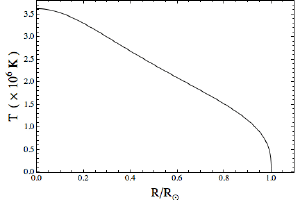
In the figures here I’ve graphed the pressure and temperature of the Sun as a function of its radius. All I’ve assumed is that I have a mass of ideal gas equal to the mass of the Sun, held together by its own weight. In astrophysics lingo, this model is one example of what’s known as a polytropic star. The exact shape of the graphs depends on the type of ideal gas you choose, so for these graphs I chose a gas that’s 75% hydrogen and 25% helium, which is roughly the percentages of the Sun.
 Brian Koberlein
Brian KoberleinThe result is that we calculate a central pressure of about 2,500 gigapascal, and a central temperature of about 3.5 million Kelvin. Compared to accepted values, our pressure is too low by a factor of about 100, and our temperature too low by about a factor of 5. Not too bad for such a rough approximation.
That said, even our rough calculation tells us something important. At the pressure and temperature we calculated it is possible for fusion to occur. Our back of the envelope calculation tells us that we should expect the core of our sun to undergo fusion. When this model was first calculated, it was a good first step to understanding what powers the Sun, and it pointed us in the right direction to develop better models.
Sometimes even a simplistic model can be very useful.