Blog
Wearing of the Green
6 January 2022
 John Vermette
John VermetteGreen is an unusual color in astronomy. It is the color to which our eyes are most sensitive, and yet few things in the night sky actually appear green. There are, for example, no green stars, only yellow-white, red, and blue ones. But there can be green comets, and we are still learning why.
 NMM London
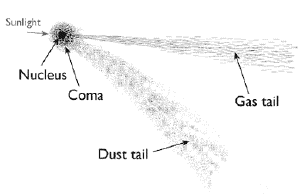
NMM LondonComets can appear green because of the chemistry of the gases vented by the comet. When a comet nears the Sun, ice on the comet’s surface sublimes, forming a halo of gas known as the coma. The solar wind pushes some of the gas away from the comet, causing it to stream out as a long tail. Most of the gas vented is water, but other compounds such as cyanide (CN2) and diatomic carbon (C2) can also be emitted. When ionized, these gasses emit a bright green glow, which can make the coma appear green.
But while the coma can appear green, even to the naked eye, the tail doesn’t. Which is odd. If the tail is just ionized gas streamed off the coma, shouldn’t it appear green as well? It’s a mystery that has puzzled astronomers for nearly a century, and a new study has figured it out.1
The longstanding idea is that over time these compounds are broken apart by ultraviolet radiation. By the time they reach the tail the molecules have been broken down to atoms, thus no green color. But proving this is extremely difficult. Diatomic carbon, which is the most common source of the green glow, is extremely reactive with oxygen and nitrogen gas, so it’s unstable in a typical lab.
To study C2, the team used a vacuum chamber, exposed C2Cl4 to a powerful laser to break it apart, then vented the chlorine gas to leave diatomic carbon. It took the team months to perfect this method, but once they had a ready supply of C2, they were able to study it in detail. They found that diatomic carbon is indeed broken apart by solar levels of ultraviolet radiation within the vacuum of space. The rate at which C2 breaks apart also helps explain why the green glow of a comet’s coma fades as the comet gets closer to the Sun.
Other astrochemistry studies show that diatomic carbon isn’t widely present on the surface of a comet. Instead, the molecule is formed through chemical reactions triggered by sunlight. So as a comet nears the Sun it gradually forms a greenish glow. The green color of a comet is short-lived, and appears only as it passes near the Sun.
Borsovszky, Jasmin, et al. “Photodissociation of dicarbon: How nature breaks an unusual multiple bond.” Proceedings of the National Academy of Sciences 118.52 (2021). ↩︎