Blog
Stone and Sea
16 November 2022
 Reto Stöckli, Robert Simmon, MODIS/NASA
Reto Stöckli, Robert Simmon, MODIS/NASAEarth is a strange world. A warm, rocky planet covered with oceans of liquid water. This strangeness is central to life on Earth, but it has been a longstanding puzzle for astronomers. Just why is our planet wet while other terrestrial worlds are dry? Where did all of Earth’s water come from?
By itself, water is an extremely common molecule. It can be found throughout the universe, and it’s plentiful on most small worlds, usually in the form of ice. Even liquid water is common. The oceans of the Jovian moons Europa and Ganymede are more vast than those of Earth. What makes Earth unusual is that it has so much water while being so close to the Sun.
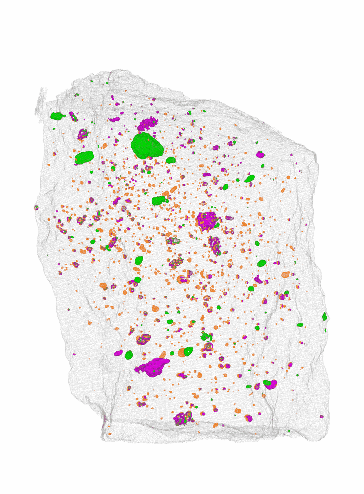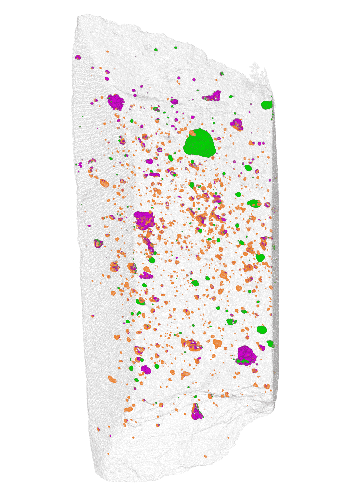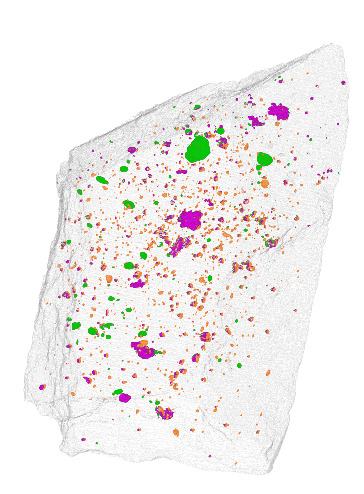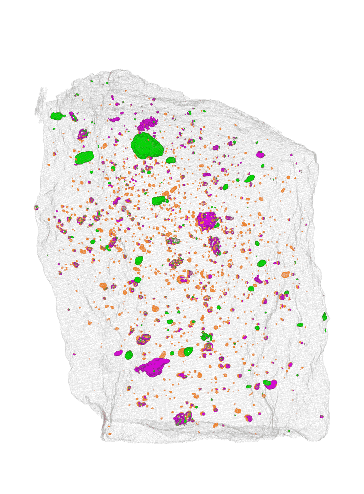 NIST
NISTBecause water is so common, planets such as Earth, Venus, and Mars were formed from water-rich dust and rocks. As the young planets settled, water was rich in their early atmospheres. But much of that water evaporated into space before the planet could completely cool. Whether an inner planet could cool fast enough to build up water on its surface is a matter of some debate. One study from Nature argues that it’s a matter of distance from its star, with Venus being too close to the Sun to cool in time, while Earth and Mars cooled soon enough.1 The idea here is that Mars then lost much of its water due to its lack of a strong magnetic field.
Another idea is that planets like Earth boiled off much of their original water, but were then seeded by water from comets and asteroids that bombarded it later. Supporters of this model point to a variance of molecular isotopes within Earth’s water. Common water is made of an oxygen atom and two hydrogen, but there is a rare variant of water that has an atom of deuterium instead of hydrogen. Only about 26 out of a million atoms are deuterium instead of hydrogen, but about 150 out of a million water molecules on Earth are the deuterium variant.
This is a key because deuterium water is much more likely to have formed in deep space. The idea is that early water in the solar system was bound to asteroids and comets, and then enriched the isotope levels of Earth’s seas. But proving that is difficult. A relatively small number of meteorites have been broken apart and analyzed at their basic molecular level. It would be difficult to do that for every asteroid we find to see if they have the right composition to explain Earth’s water. But a new study has found a way to make it easier.2
The team at NIST found that instead of cutting off slices of a meteorite to analyze it chemically, they could scan it using a combination of x-ray and neutron imaging. To prove its accuracy, the team scanned two meteorites that had already been chemically analyzed. They compared their scans to the known composition, and they matched quite well.
One big advantage of this new method is that it’s non-destructive. The scans don’t damage the meteorites in any way. The scans also yield a 3D image of where water and other compounds are located within the rock, unlike chemical methods which only confirm the presence of these compounds.
In the future, the team hopes to use this method on many more meteorites, which could pin down the chemistry of early asteroids and help solve the mystery of Earth’s water. It could even determine other compounds brought to Earth, such as the building blocks of life itself.
Hamano, Keiko, Yutaka Abe, and Hidenori Genda. “Emergence of two types of terrestrial planet on solidification of magma ocean.” Nature 497.7451 (2013): 607-610. ↩︎
Treiman, Allan H., et al. “Coordinated neutron and X‐ray computed tomography of meteorites: Detection and distribution of hydrogen‐bearing materials.” Meteoritics & Planetary Science 57.10 (2022): 1820-1835. ↩︎