Blog
Making Waves
2 May 2014
 KEK
KEKGeorge Thomson had some big shoes to fill. His father J. J. Thomson had won the Nobel prize for the discovery of the subatomic particle now known as the electron, and George had become a physicist as well. Fortunately George Thomson did quite well for himself, and was also awarded a Nobel prize. In a way, George won his Nobel prize for proving his father wrong.
 Wikipedia user Kurzon
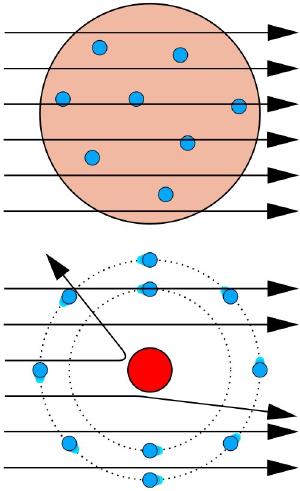
Wikipedia user KurzonWith J. J. Thomson’s discovery of electrons, there was a strong effort to determine the structure of atoms. One of the more popular ideas was known as the plum pudding model, which proposed that atoms were basically a sphere of positive charge embedded with negatively charged electrons, like plums in a pudding. But experiments soon showed this model wasn’t correct. When gold foil was bombarded with alpha particles (what we now know are high energy helium nuclei) some of them were scattered at large angles, which indicated that most of the atomic mass must be concentrated in a nucleus. So a different model was proposed, similar to that of the solar system. In this model, the positively charged nucleus formed the center of the atom, and the electrons orbited it much like the planets orbit the Sun.
But there is a major problem with the solar system model of the atom. If the electrons orbited the nucleus like a planet, their electric charge would produce electromagnetic waves that would radiate away from the atom. As a result, the electron would lose energy and spiral into the nucleus. A basic calculation shows that this would occur within a fraction of a second. So a consequence of this model is that atoms are unstable, when clearly many atoms are quite stable.
To solve this problem Niels Bohr proposed that rather than orbiting the nucleus, electrons were quantized into discrete orbitals. This quantization meant that electrons couldn’t collapse into the nucleus, thus making atoms stable. One way to interpret this idea is to propose that electrons are a matter wave, similar to the way light is a wave. The idea of matter waves was first proposed by Louis de Broglie, and if true it would mean that even the free electrons of the cathode ray tubes should have a wavelength.
 Lambda Scientific Systems
Lambda Scientific SystemsIt was an interesting idea, but the theoretical wavelength of an electron was extremely small, about the same as the diameter of an atom. How could one possibly measure such a wavelength? George Thomson found the solution using a cathode ray tube similar to his father’s. In George’s device, a beam of electrons was aimed at a crystalline material, behind which was a phosphor screen that would light up wherever an electron struck it. If the the electrons were particles, then they would strike the atoms in the crystalline material and scatter in all directions. Thus there would be an even distribution of electrons on the screen and the screen should have a fairly uniform glow. If, however, the electrons were waves, then the crystalline material would cause ripples in the electron beam. These ripples would interfere with each other, similar to ripples on a pond. These interfering ripples would then create a kind of ripple pattern on the screen.
When George Thomson did this experiment, he found that a rippled pattern did indeed form on the screen. By measuring the spacing of the ripples, Thomson was able to determine the wavelengths of the electrons, and they agreed with de Broglie’s theory. Electrons were indeed waves. A similar experiment was done independently by Clinton Davisson and Lester Germer. In 1937 Thomson and Davisson were awarded the Nobel Prize in Physics for their demonstration of the wave nature of electrons.
So Thomson the senior demonstrated that electrons were particles, and Thomson the junior demonstrated that they were waves. These seemingly contradictory discoveries were two of several that led to the development of quantum theory, which revolutionized our understanding of the universe.