Blog
Chain Reaction
10 June 2014
It is often said that we are made of star stuff. Except for hydrogen, the atoms in our bodies were fused in supernovae, and in the cores of stars. What’s not often talked about is just how complex fusion is, and how difficult it is to do, even in the core of a star. Take, for example, the seemingly simple fusion of hydrogen into helium, which is the primary energy source of our Sun.
A simple calculation of the Sun’s interior puts the temperature of the Sun’s core at about 3 million Kelvin. It is actually closer to 15 million Kelvin, and has a pressure more than 300 billion times that of Earth’s atmosphere at sea level. At such a high temperature and pressure, you can imagine that hydrogen nuclei are slamming into each other all the time. But simply having nuclei collide is not enough to make them fuse. Hydrogen nuclei (protons) are positively charged, so the closer they get the harder they push each other away. This repulsive force between nuclei is so strong that most collisions aren’t strong enough to overcome it.
 ATNF
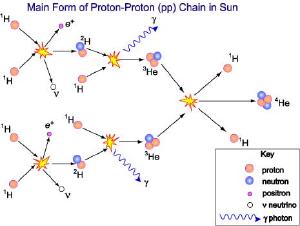
ATNFFortunately, they don’t have to overcome all the repulsive force. Through an effect known as quantum tunneling, nuclei that get almost close enough can rely on quantum mechanics to get the rest of the way there. Not all close collisions result in quantum tunneling, but enough do that protons can fuse into helium 2 (also known as a diproton). Unfortunately, most of these helium 2 nuclei immediately decays into two protons. Unlike the more common helium 4, helium 2 is incredibly unstable, so simple proton collisions do not produce stable helium. However, on rare occasions helium 2 will decay into deuterium, plus a positron and neutrino. The neutrino quickly escapes the Sun, and the positron typically collides with an electron to produce gamma rays.
A deuterium nuclei consists of a proton and neutron, and is a stable isotope of hydrogen. So even though the formation of deuterium from proton collisions is exceedingly rare, deuterium can build up in the solar core. Usually deuterium will collide with a wandering proton to create helium 3. Helium 3 is a stable isotope, and thus can also build up in the Sun’s core.
Because helium 3 has two protons, it is very difficult for a proton to collide with helium 3 to produce lithium 4. Lithium 4 is also extremely unstable, so such a reaction wouldn’t be very useful. Instead, what typically happens is a helium 3 nuclei will collide with another helium 3 to produce beryllium 6, which quickly decays into helium 4 and two hydrogen nuclei.
This process of protons to deuterium to helium 3 to helium 4 is one example of what is known as the proton-proton chain (or pp-chain). It is the primary source of energy produced within the Sun’s core. There are other pp chains that are similar, as well as other fusion processes such as the CNO cycle that contribute to solar fusion. In larger stars there are further nuclear interactions that produce heavier elements.
So it is true that we are made of star stuff, but the process of making that star stuff takes some fairly complex nuclear physics.